 
Always check the results rounding errors may occur. Thus there are 1000 or 10 3 milliliters in a liter. #GRAMS TO LITERS CHEMISTRY CALCULATOR HOW TO#Use this page to learn how to convert between liters and grams. Liter, on the other hand, is a measure of volume (container capacity). Multiply the normality by the mass equivalent and the volume of the solution (in L) to calculate mass (in grams) of the dissolved compound. The basic unit of measurement for mass in the metric system one cubic centimeter of water has a mass of approximately one gram. 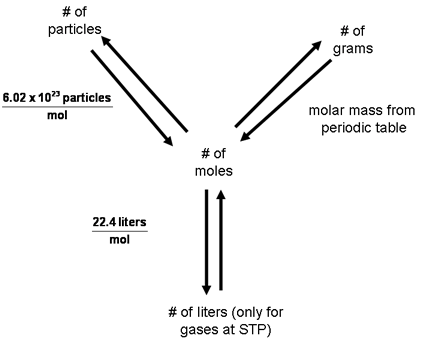
How to do Mole Conversions: At first, use the mole map to set up and solve problems. When multiplying a decimal by 10, move the decimal point one place to the left. We begin by comparing the organic compounds to compounds that do and do not include carbon. Always plan problems so you can cancel units. As usual, we just set it up so that the starting units cancel and we end up with the unit we. 1 Grams = 0.001 liter 1 Liter = 1000 Grams Grams To Liters 1 liter (l) = 1000 gram (g). How many moles of water molecules, H 2 O, are present in a 27 gram sample of water? In a general chemistry class, you usually end up having to perform a lot of conversions involving moles (mol). In many chemistry problems, you need to convert grams to moles or moles to grams. For example, gold's density is 19.3 grams per mL. 37 Liter to Gram Conversion 38 Grams, Kilograms, and Liters How Many Grams Is A Liter? The below formulas only offer a generalized equation: 1 liter is approximately equal to 2.2 pounds. So 3.5 moles should be about three times. Example: How many moles of sodium are there in a 0.59 gram sample of sodium metal. you have moles per liter If you have moles and know the formula weight of the compound you can solve for grams Moles = gr / formula weight Once you have grams you can get grams / liter. Answer 1.74 L Of course, once quantities in moles are available, another conversion can give the mass of the substance, using molar mass as a conversion factor. 26 Votes) This is the weight in grams of 1 mole of the compound. 
If starting with grams, we use 1 mL/19.3g to convert to milliliters. Use our liters to kg calculator to find how many kilograms in a liter. Reference data: Density of oxygen = 1.429 kg/m 3 (for 25 C) Mass (weight) and volume depend on temperature, pressure and composition due to density changes. Chemical Formula : (Optional) Molar Mass: Mass Concentration. The below formulas only offer a generalized equation: 1 liter is approximately equal to 1000 grams. Liter ( l) is a unit of Volume used in the Metric system. mass = d × v × vcf mcf, where vcf is the conversion factor to convert from liter to cubic meters. C 4 quarts = 1 gallon, 16 quarts = 16/4 = 4 gallons. From particles (atoms, molecules, or formula units) to moles: Divide your particle value by Avogadro's number, 6.02 × 10 23. The density of liquid water is 1000 grams / liter. Calcium, for example, has an atomic mass of 40. 
#GRAMS TO LITERS CHEMISTRY CALCULATOR FREE#That is why students and professionals make a vast use of this free grams to moles calculator for accurate outputs so as to avoid any hurdle during the reaction.When you denote the concentration of the solution using normality, it means you are expressing the weight of the solute in its gram equivalent per liter of the final solution. Moreover, it also gives an idea about the elements that are involved in a particular chemical reaction. We convert the given mass of any compound or substance to its corresponding number of moles because it allows us to write the simplest chemical formula of the substance. If we have 5g of HCl, then we can say that it has 0.137 moles. The number of moles in 1 gram of HCl is 36.461. Let us go through the organic content below to understand how to use this free calculator.īasically, mole is the SI unit of the substance quantity.ġ mole of any compound or substance contains exactly \(6.02214076 * 10^ = 1 Mole $$ How many moles are there in 5g of an element?įor instance, let us talke an example here. An online grams to moles calculator is exclusively programmed to convert grams to moles, moles to grams, molar mass and number of the molecules present in the substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
